CDISC SEND Services
SEND conversion, validation, and submission-ready support for nonclinical toxicology and preclinical study data.


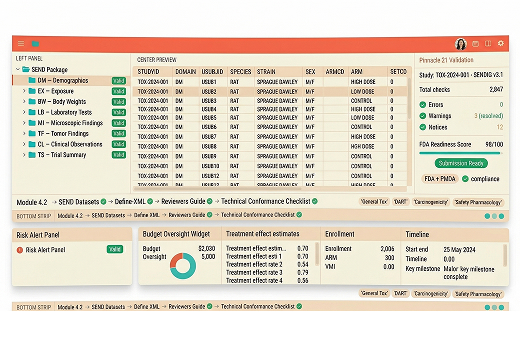
From Raw Nonclinical Data to Submission-Ready SEND
Inductive Quotient converts raw nonclinical study data from platforms such as Provantis, Pristima, Watson LIMS, spreadsheets, and legacy sources into structured, SEND-compliant datasets built for regulatory submission. We support the full SEND lifecycle from source assessment and domain mapping through controlled terminology, validation, Define-XML, and reviewer-ready submission assets.
Our team works across SEND implementation guide classes, including Findings, Events, Interventions, and Trial Design, with a delivery model built for traceability, consistency, and quality control. For multi-CRO and multi-lab programs, we help standardize data across systems, species, and study designs so sponsors can assemble a cleaner, more submission-ready nonclinical portfolio.
Regulatory Readiness
End-to-End SEND Expertise
From Raw Data to Submission
Let Us Prepare Your SEND Submission
Get your SEND package right the first time - source data assessment to submission-ready datasets.
Talk to Us