PK/PD Analysis & Pharmacometrics
Noncompartmental, compartmental, and population PK/PD analysis to characterize exposure, understand response, and support dose and study decisions across preclinical development.


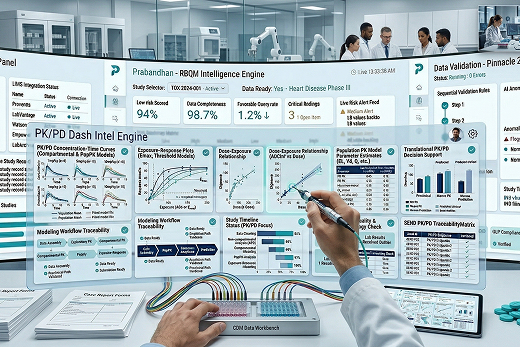
Quantitative PK/PD Insight for Better Development Decisions
PK/PD and Pharmacometrics translate concentration-time data into development decisions. We use noncompartmental analysis, compartmental modeling, and population PK/PD approaches to characterize exposure, understand response, and support dose selection across preclinical programs.
Our work connects bioanalytical data, toxicology findings, and pharmacology signals into one quantitative framework. This helps sponsors evaluate exposure-response relationships, compare species, project first-in-human starting doses, and strengthen model-informed decisions with traceable, scientifically grounded analysis.
By combining regulatory expertise, market intelligence, and structured submission planning, we help manufacturers move from early regulatory assessment to submission-ready dossiers for FDA, EU MDR/IVDR, PMDA, CDSCO, LATAM, and other global markets.
Core Elements of Our Approach
Six Modeling Disciplines. One Integrated Practice.
Covering the full pharmacometric spectrum from NCA through MIDD.
Six Steps from Data to Insight
A structured pharmacometric workflow from raw data through regulatory-ready deliverables.
Ingest
Aggregation of concentration and biomarker data from bioanalytical labs.
Explore
Exploratory data analysis (EDA) to identify trends and outliers.
Model
Structural model development and parameter estimation.
Validate
Goodness-of-fit assessments and visual predictive checks (VPC).
Simulate
Trial simulations to predict outcomes under different dosing scenarios.
Report
Final modeling report delivery for regulatory submission.
Software Platforms and Standards
Optimize Your Development Strategy
Ensure your dose selection is backed by rigorous pharmacometric modeling.
