CDQOps
AI-Powered Clinical Data Quality Automation.


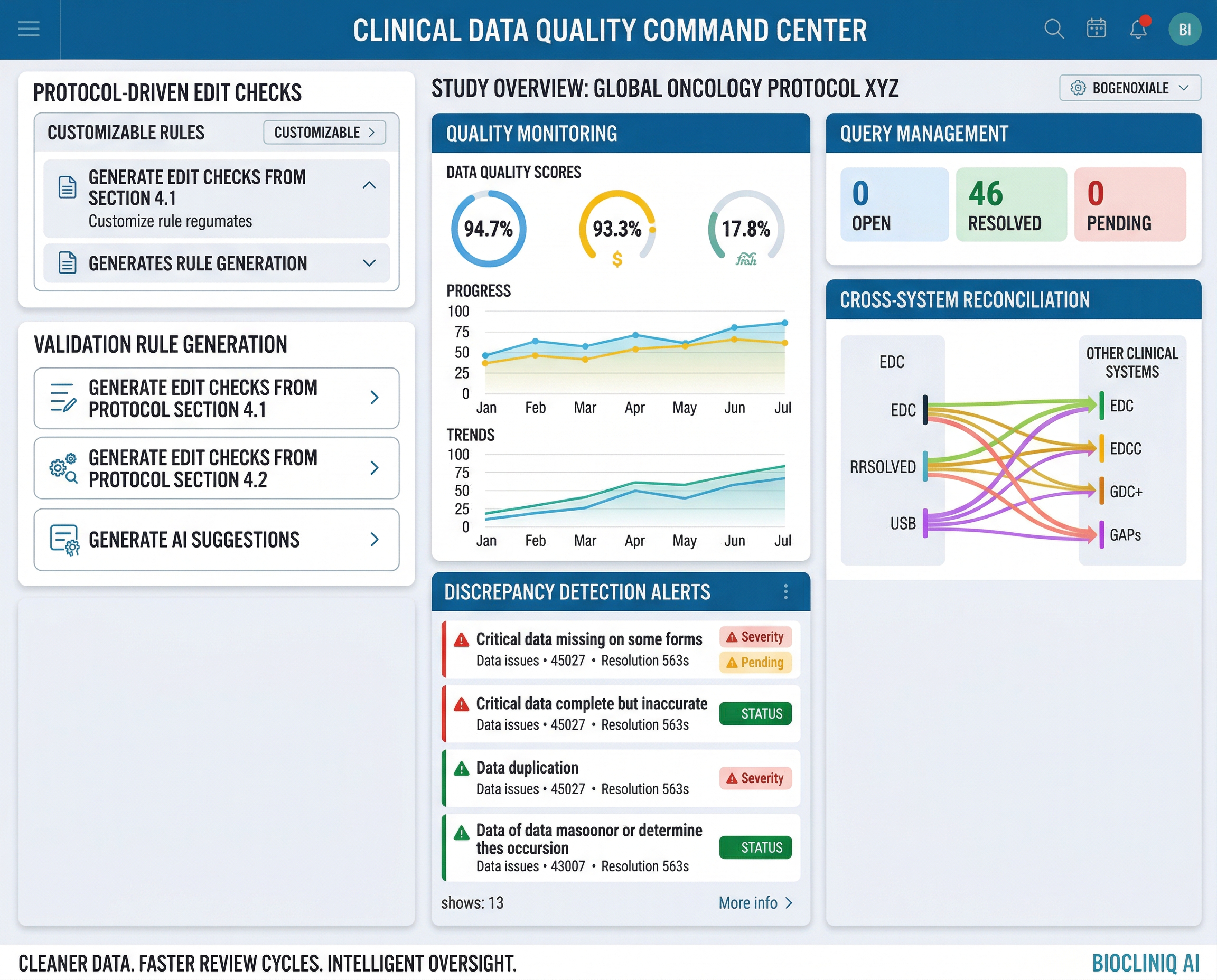
Overview
CDQOps is Inductive Quotient’s AI-powered clinical data quality and validation platform designed to improve the speed, consistency, and control of study data review. It helps teams identify data issues early, automate validation workflows, and strengthen oversight across clinical data management activities.
Built to support modern study execution, CDQOps streamlines edit check management, discrepancy identification, reconciliation support, and data review workflows through intelligent automation and configurable quality rules. By reducing manual effort and improving traceability, it helps clinical teams accelerate data cleaning, improve data reliability, and move faster toward database lock.
Built to automate clinical data quality workflows across validation, reconciliation, and review.
Why IQA CDQOps?
Built to reduce manual effort, improve consistency, and accelerate high-quality clinical data review.
Faster Study Timelines
- Accelerate validation, reconciliation, and review workflows to move studies forward with cleaner data and less manual effort.
Accuracy and Consistency by Design
- Apply standardized validation, automated checks, and unified quality logic to improve consistency across studies, sites, and systems.
Flexible for Complex Study Environments
- Adapt to different study designs, rules, and workflows with scalable automation built for evolving data quality needs.
Multi-EDC Compatibility
- Work across major EDC platforms through a flexible connector framework that supports consistent quality operations across environments.
Greater Operational Efficiency
- Reduce manual programming, repetitive review cycles, and data management burden through more automated quality workflows.
Quality Scoring Dimensions
Five dimensions that shape the overall clinical data quality score
Completeness
Measure required data population across forms, visits, and study workflows.
Accuracy
Evaluate whether data values align with expected ranges, formats, and business rules.
Consistency
Identify mismatches across forms, visits, and connected data sources.
Timeliness
Track data entry, query response, and review turnaround across study activities.
Conformance
Assess alignment with standards, controlled terminology, and coding requirements.
Ready to Automate Your Data Quality Operations?
See CDQOps generate edit checks from protocol text and auto-resolve queries.
