Purpose-Built Solutions for Modern Life Sciences & Healthcare
Overview
Connected Solutions Built for Real Life Sciences and Healthcare Work
IQA’s solutions combine domain expertise, product thinking, and AI-driven innovation to support the workflows that matter most across clinical development, regulatory operations, data quality, oversight, coding, and real-world evidence. These are not generic technology tools. They are purpose-built platforms and AI copilots designed to work in regulated, data-intensive, and operationally critical environments.
From study build and clinical oversight to protocol intelligence, medical coding, data quality operations, and evidence generation, our solutions help organizations improve speed, visibility, traceability, and execution across the full life sciences and healthcare value chain.
Core Solutions
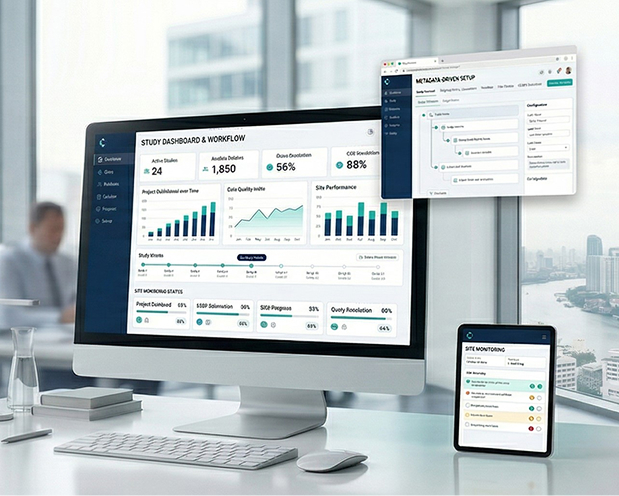
InductiveEDC
A metadata-driven EDC platform built to support faster study build, cleaner data capture, stronger oversight, and more efficient downstream execution across clinical research programs.

RBQM
A risk-based quality management solution that transforms clinical and operational data into actionable oversight, helping teams identify issues earlier and manage trial risk more proactively.
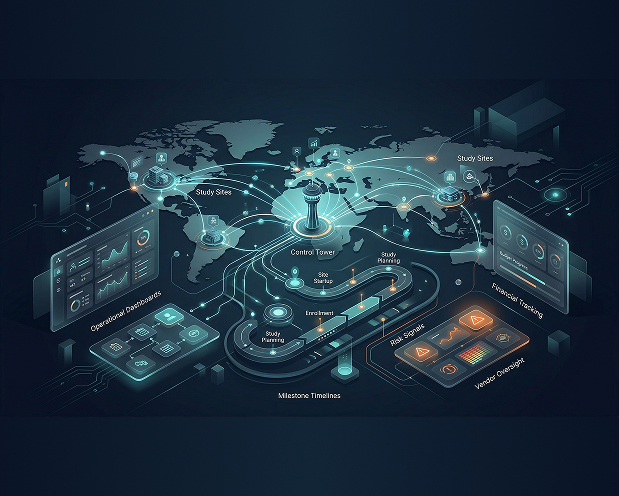
Prabandhan
A unified project oversight and meta-analysis solution that brings together study operations, milestones, vendors, compliance, financial tracking, and cross-study visibility in one connected environment.
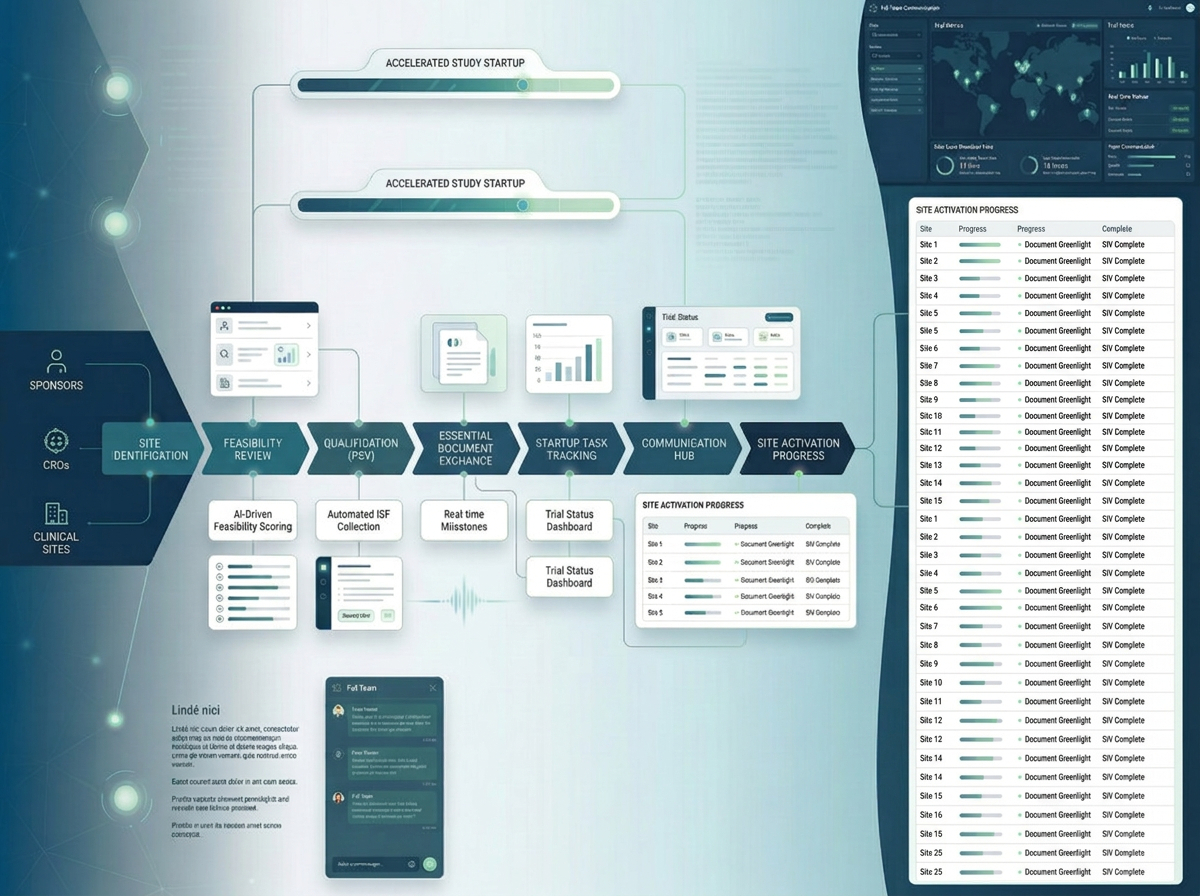
TrialLattice
A collaborative platform designed to streamline study start-up, strengthen site and stakeholder coordination, and connect workflows across study teams and partners.

ClinEvra
A metadata-driven, end-to-end workflow orchestration solution that dynamically extracts study assets from protocols and transforms them into structured, downstream-ready outputs for database build, validation, trial domains, SDTM preparation, and regulated study execution.
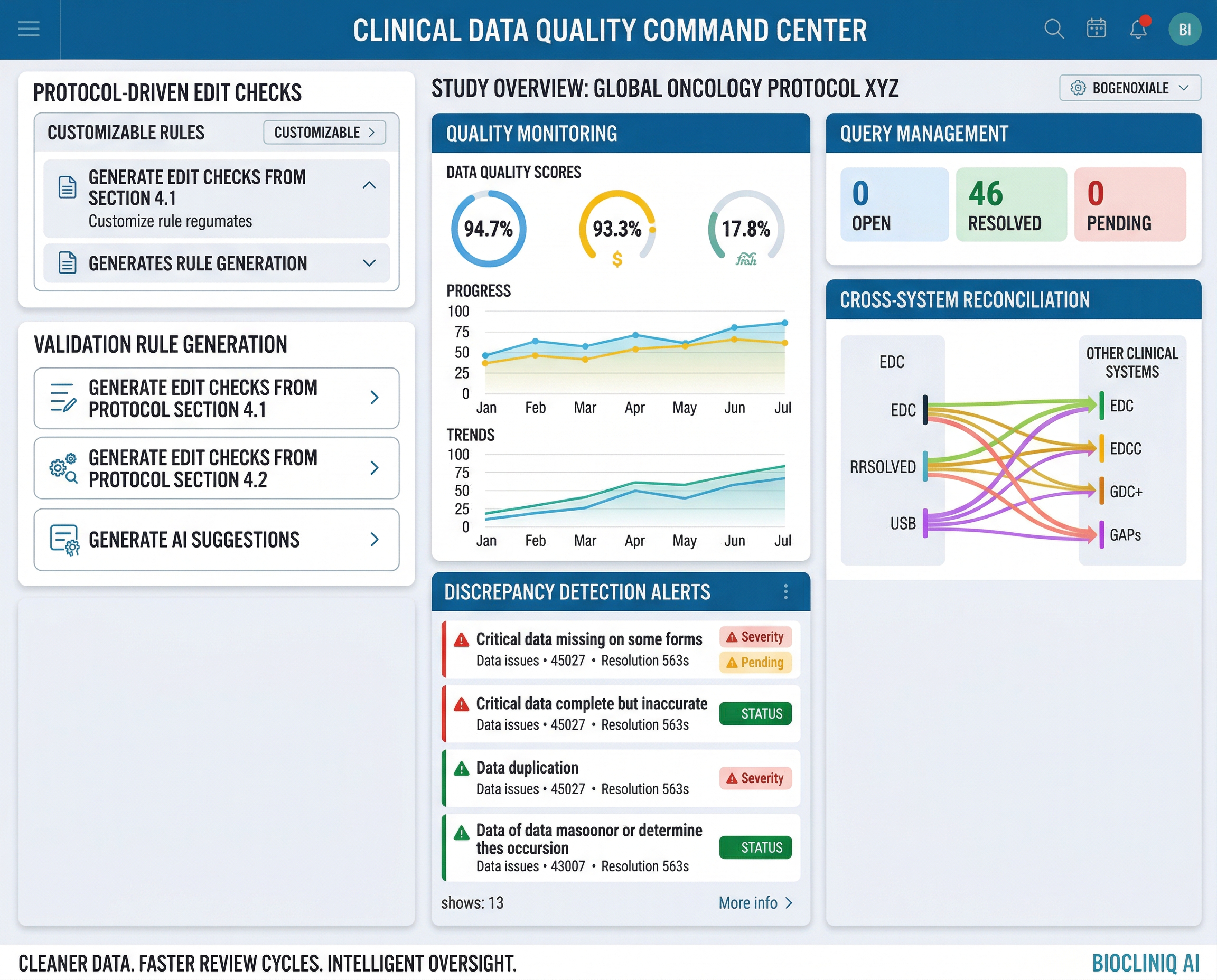
CDQOps
A quality-by-design data operations solution that improves validation, review, and oversight across clinical data workflows through traceable issue management, consistency controls, and operational visibility.
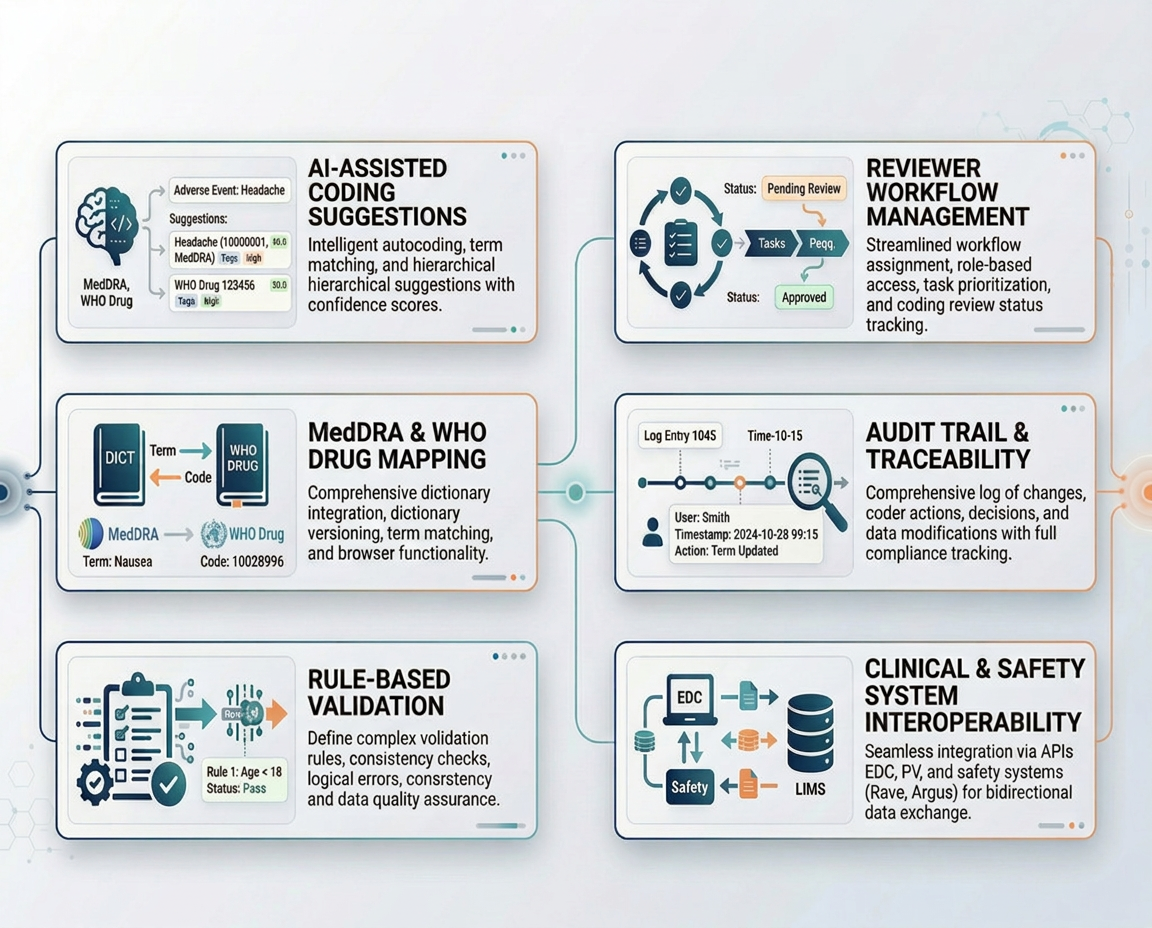
Aushadhi
An AI-assisted medical coding platform that streamlines coding workflows, improves consistency, and supports traceable human review across clinical trial and safety workflows.

Evidexia
A platform for real-world evidence generation that supports data integration, curation, analytics, and evidence development across real-world, observational, and post-market environments.
Ready to Transform Your Clinical Operations?
Schedule a personalized demo of any solution or discuss a custom platform configuration for your organization.
