Prabandhan
Gain end-to-end visibility across clinical programs with one connected platform.


Built for Unified Clinical Oversight
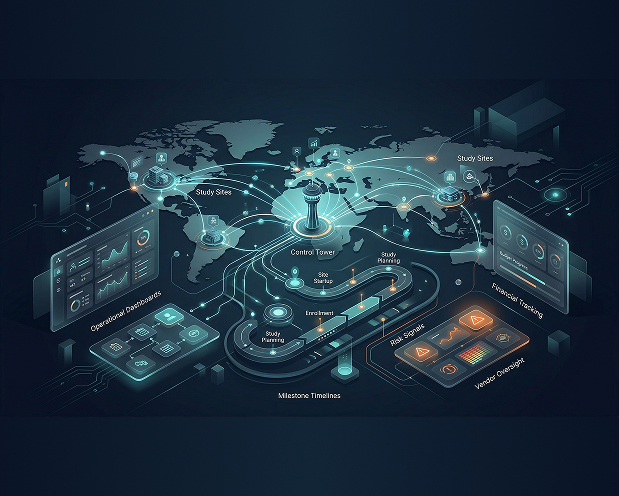
Prabandhan is Inductive Quotient’s unified oversight platform for clinical programs, designed to bring study, vendor, regulatory, milestone, and financial tracking into one connected environment. It helps teams manage operational complexity across studies with better visibility, stronger coordination, and more proactive control.
The platform also extends into advanced use cases such as meta-analysis across studies, including PK/PD parameter analysis, while Agentic AI capabilities help extract and dynamically populate study-level information to accelerate execution and improve consistency.
Built for Study, Vendor, and Regulatory Oversight
Study Lifecycle Management
Prabandhan manages every phase from registration through submission.
Study Planning
Define protocol milestones, create budgets, model enrollment scenarios and allocate resources.
Site Activation
Track feasibility, contracts, regulatory submissions and site initiation across all countries.
Trial Execution
Monitor enrollment, manage CRA visits, track vendors and control budgets in real-time.
Submission and Disposal
streamlines regulatory submissions and ensures timely, compliant handling of approvals, closures, and document lifecycle management.
Ready to Optimize Your Trial Management?
See Prabandhan's AI planning and financial management in a personalized demo.